The Torah portion Tzav describes a ritual performed by Moses in consecrating Aaron as the High Priest (Kohen Gadol) and his sons as priests (kohanim):
And the other ram was presented, the ram of consecration, and Aaron and his sons laid their hands upon the head of the ram. And when it was slain, Moses took of the blood thereof, and put it upon the tip of Aaron’s right ear, and upon the thumb of his right hand, and upon the great toe of his right foot. And Aaron’s sons were brought, and Moses put of the blood upon the tip of their right ear, and upon the thumb of their right hand, and upon the great toe of their right foot; and Moses dashed the blood against the altar round about.
Leviticus 8:22-24
This narrative always struck me as one of the most mysterious rituals in the Torah. What could be the meaning of putting sacrificial blood on the right earlobes, the right thumbs, and the right big toes of Aaron and his son? It sounds so random. Rashi is silent on these verses, and other classical commentators offer little help in understanding this mystery.[1]
However, three scientific parallels—one from biochemistry and two from physics—will help unravel this mystery.
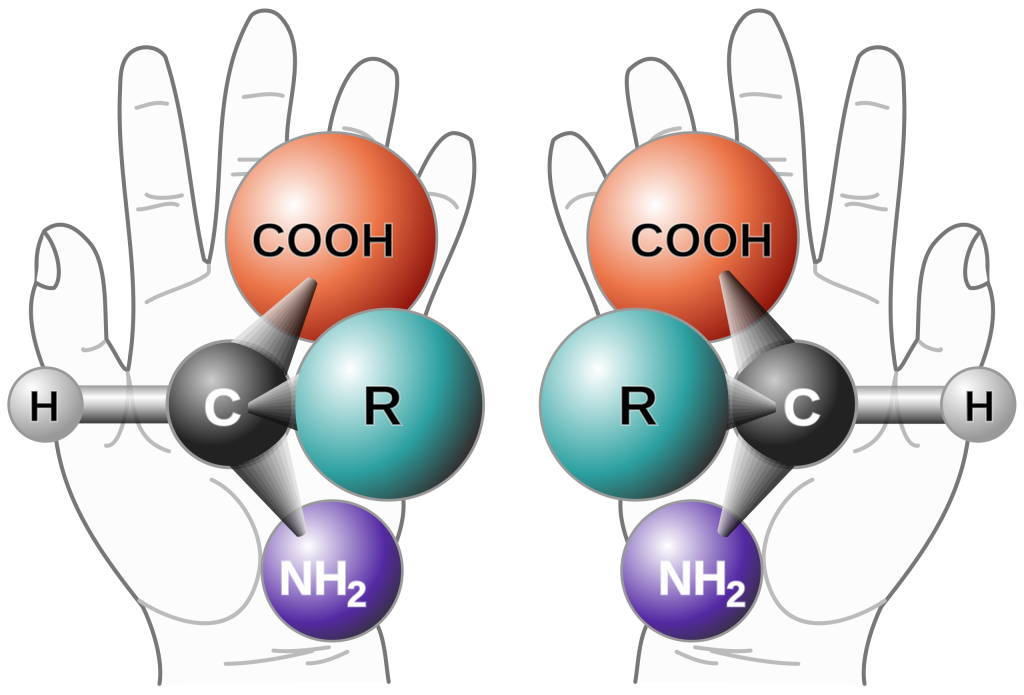
Figure 1. Two enantiomers of a generic amino acid that are chiral. Public Domain
Chirality, also known as handedness, is a fundamental concept in organic chemistry that refers to the property of an object or a molecule to exist in two non-superimposable mirror-image forms. Just like your left and right hands are mirror images of each other but cannot be perfectly aligned (or superimposed) on top of one another, chiral molecules and their mirror images cannot be superimposed. This phenomenon is a source of fascination and mystery in organic chemistry.[2]
The puzzle of chirality arises from the fact that two mirror-image forms of a chiral molecule, called enantiomers, have identical chemical and physical properties in an achiral environment (an environment that is not itself chiral). However, these enantiomers exhibit different properties and behaviors when placed in a chiral environment, such as in the presence of other chiral molecules or a magnetic field.
This phenomenon has several profound implications. Many biomolecules, such as amino acids, sugars, and proteins, are chiral. Enantiomers of chiral drugs or biomolecules often have drastically different biological activities, with one enantiomer being therapeutically effective while the other may be inactive or harmful.
One of the distinguishing characteristics of chiral molecules is their ability to rotate plane-polarized light, a property known as optical activity. One enantiomer will rotate the plane of polarization in one direction (dextrorotary, or “right-handed”), while its mirror image will rotate it in the opposite direction (levorotary, or “left-handed”).
A racemic mixture (or racemate) is a 50:50 mixture of two enantiomers of a chiral molecule. Because the optical activities of the enantiomers cancel each other out, racemic mixtures are optically inactive. Separating enantiomers to get one specific enantiomer is often necessary for pharmaceutical applications, as the biological effects of the enantiomers can be very different.
Chirality—right-handedness or left-handedness—is a fundamental aspect of molecular geometry that plays a critical role in the physical and chemical properties of compounds, especially in how they interact with biological systems. Importantly, chirality symmetry dictates that, as a result of chemical reaction, there should be exactly 50% of left-handed molecules (L-amino acids) and 50% of right-handed molecules (D-amino acids). However, in living organisms, almost all amino acids used to build proteins are left-handed—L-enantiomers. This violation of symmetry is a great mystery of biochemistry.[3]
This asymmetry is not incidental. If not for the left-handed asymmetry of amino acids, life would not be possible. Proteins are built from chains of amino acids. Each amino acid has a specific 3D structure due to its chirality. If a protein is made with D-amino acids (right-handed) instead of L-amino acids (left-handed), it would fold differently and not function properly.
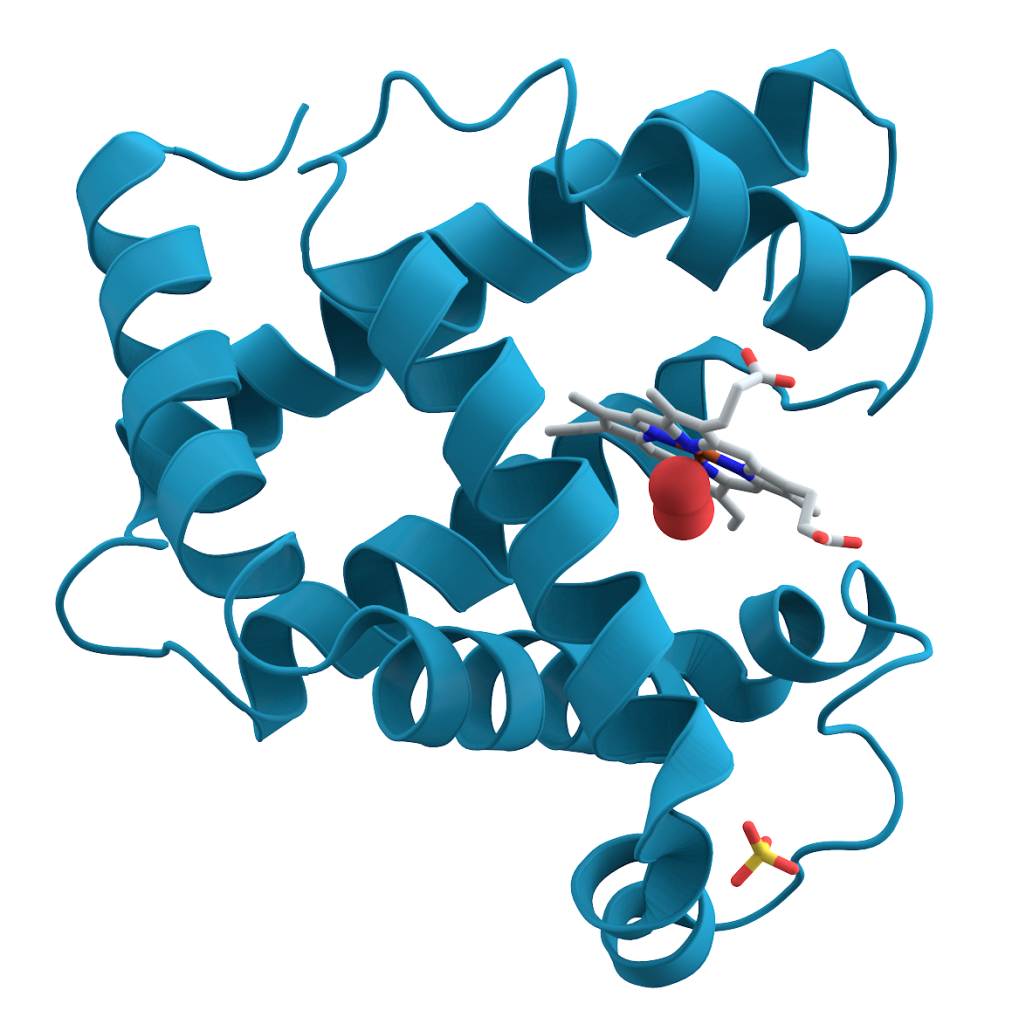
L-amino acids “fit” together to create the complex and functional shapes of proteins essential for life. D-amino acids, on the other hand, would not fit into these protein structures, disrupting vital biological processes. D-amino acids can be found in the cell walls of some bacteria and may play a role in specific functions like antibiotic resistance.[4]
In living organisms, the sugars used for energy production (like glucose) and structural components (like the sugar backbone of DNA) are almost exclusively right-handed (D-glucose, D-deoxyribose). This preference, like with amino acids, is crucial for proper function within the biological machinery. (Although rare, some bacteria can utilize L-sugars for specific purposes.)
The dominance of L-amino acids and D-sugars in life is a fascinating example of how seemingly random symmetry breaking profoundly impacts biological systems. The specific 3D structure of these molecules is crucial for protein function, and life, as we know, depends on this “left-handedness.” This is a great example of the “fine-tuning” of physics and chemistry to allow for life.
The mysterious asymmetry between matter and antimatter[5] in the universe presents another intriguing example of fine-tuning. The Standard Model, our most comprehensive theory of subatomic particles and their interactions, predicts that the Big Bang should have created equal amounts of matter and antimatter. However, these particles annihilate each other upon contact, releasing pure energy. If this prediction were true, all matter and antimatter would have vanished, leaving a universe filled only with light. Fortunately, this isn’t the case—the vast majority of matter in the universe lacks an antimatter counterpart. Scientists are still searching for the reason behind this imbalance, a phenomenon known as the baryon asymmetry problem. Whatever the reason, we owe our existence to this lucky imbalance.
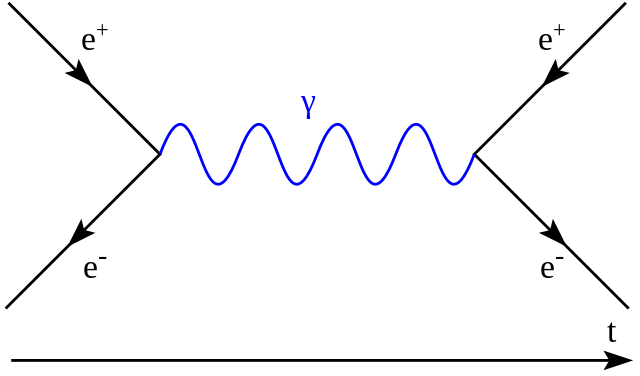
A Feynman diagram showing the annihilation of an electron and a positron (antielectron), creating a photon that later decays into a new electron–positron pair.
This asymmetry shares some similarities with the dominance of left-handed L-amino acids in biological life. Both phenomena represent a form of “fine-tuning,” where the universe seems to have specific properties that favor the existence of complex structures like stars, planets, and life itself.
One can draw an interesting parallel between the L-amino acids and D-sugars on the one hand and sefirot (divine emanations or lights—from the Hebrew word sapir—to “shine”) on another. The two main (and opposite) sefirot are ḥesed and gevurah (which respectively represent thesis and antithesis). In the sefirotic tree Etz Ḥayim, ḥesed represents the right column, and gevurah represents the left column. Ḥesed is sweet (its role is to sweeten judgments), and gevurah (judgment) is bitter.
Interestingly, most right-handed sugars are sweet. Many common sugars we encounter, like glucose, fructose, and sucrose (table sugar), are D-sugars, e.g., the right-handed sugars.[6] On the other hand, most left-handed amino acids are bitter. Common amino acids such as leucine isoleucine, valine, arginine, methionine, phenylalanine, tryptophan, and histidine are tasteless or have a bitter taste.[7] As we see, the right- or left-handedness and the sweet or bitter taste of sugars and amino acids line up with the same characteristics of sefirot—ḥesed and gevurah:
| Sefira/Molecule | Ḥesed/Sugars | Gevurah/Amino acids |
| Handedness | right-handed | left-handed |
| Taste | Sweet | bitter |
This parallel may not be co-incidental. As mentioned before, sefirot can be translated as lights. However, in a more nuanced view, each sefirah comprises the infinite divine light (Or Ayn Sof) and a vessel (keli), which give the simple light of the divine its specific characteristics. Using the physical metaphor of light polarization, we can say that the sefirot of the left column “polarize” the divine light in one plane, whereas the sefirot of the right column “polarize” the divine light in the perpendicular plane. But, discussed above, chiral molecules also rotate plane-polarized light. One enantiomer will rotate the plane of polarization in one direction (dextrorotary, or “right-handed”), while its mirror image will rotate it in the opposite direction (levorotary, or “left-handed”). As we can see, the parallel between sefirot and chiral molecules of sugars and amino acids runs deep.
Now, we can finally return to the mysterious ritual of the inauguration of priests. According to the Arizal, the Levites personify gevurah, whereas the Kohanim—the priests—personify ḥesed. However, Aaron and his sons were also Levites as they descended from the tribe of Levi. Consequently, before the inauguration, they represented the mixture of ḥesed and gevurah. Recall that a racemic mixture (or racemate) is a 50:50 mixture of two enantiomers of a chiral molecule. Using this metaphor, we can say that, before the inauguration, Aaron and his sons were a racemic mixture of ḥesed and gevurah. Because the optical activities of the enantiomers cancel each other out, racemic mixtures are optically inactive. So, too, with Aaronites, before the inauguration, their ḥesed and gevurah, which are thesis and antithesis, canceled each other out, making them spiritually inactive. And just as separating enantiomers to get one specific enantiomer is often necessary for pharmaceutical applications, to empower Aaronites to serve in the Temple, Moses had to separate ḥesed and gevurah within them. It seems that this may be the meaning of the mysterious inauguration ritual.
G‑d created a man in His image and His likeness (Genesis 1:27). Kabbalists interpret this verse to mean that the human body reflects the structure of the sefirotic tree: Keter manifest as a skull; ḥokhmah and binah—as two hemispheres of the brain; ḥesed and gevurah—as two arms; tiferet—as torso; netzaḥ and hod—as two legs, Yesod—as the procreative organ; and malḥut—as a female.
The human body is symmetrical along the vertical axis. That is, our left side is a mirror reflection of our right side. That is why, when we look in the mirror, we see ourselves not even realizing that the right and the left sides are switched in the mirror reflection. To separate ḥesed and gevurah within Aaronites, Moses had to break that symmetry. To accomplish this, Moses marked with sacrificial blood the right-most extremities of the bodies of Aaron and his sons—their right earlobe, the right thumb, and the right big toe. By consecrating the right site, Moses made Aaronites spiritually right-handed so that ḥesed became their dominant sefirah. From then on, Aaronites were no longer a racemic mixture of ḥesed and gevurah. This made them conduits of the divine light that shone through the priests onto the rest of the Israelites.[8]
This year, on the week we read the Torah portion of Tzav, we also celebrated Purim. Nothing happens coincidentally. So, what is the connection between Purim and Tzav?
As I wrote in my essay, “Purim—the Day When We Celebrate Spontaneous Symmetry Breaking,” the essence of Purim is spontaneous symmetry breaking. Hamman aimed at reaching the level where there was no difference between Jews and non-Jews. To reach this level, he cast dies. Haman succeeded in reaching the level that transcends all differences, where there is complete symmetry between right and left. What he did not realize—and got hanged due to this grave error—was that on this lofty level, the symmetry gets broken.
I loved you, said the Lord, and you said, “How have You loved us?” Was not Esau a brother to Jacob? says the Lord. And I loved Jacob. And I hated Esau…
Malachi I, 2-3
The above prophesy is a manifesto of symmetry breaking.
I see in this parallel the connection between Purim and the Torah portion of Tzav, where Moses had to break the symmetry between left and right to empower Aaronites to serve as priests in the Tabernacle.
Endnotes:
[1] Philo of Alexandria taught that this ritual signified that the perfect person must be pure in every word, action, and in all of life. For it is hearing that judges a person’s words, the hand is the symbol of action and the foot of the way in which a person walks in life. Philo taught that since each of these parts is an extremity of the body, and on the right side, this indicates that improvement in everything is to be arrived at by dexterity, being a portion of felicity, and being the true aim in life, which a person must necessarily labor to attain, and to which a person ought to refer all actions, aiming at them in life as an archer aims at a target. (Philo, On the Life of Moses 2:29:150)
[2] See National Center for Biotechnology Information (NCBI): Chirality (stereochemistry): https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5765859/) This website provides a great overview of chirality, including its definition, types (enantiomers and diastereomers), and its significance in biological molecules. See also “Biochemistry” by Lubert Stryer. This classic biochemistry textbook has a dedicated chapter on stereochemistry (Chapter 3) that covers chirality in detail, including its role in amino acids, carbohydrates, and other biomolecules.
[3] The exact reason for the dominance of L-amino acids in life is an area of active research. Several hypotheses have been put forward to explain this mystery, but all very speculative.
[4] See for example, George L. Kenyon et al., “Chirality and Its Importance in Biology,” The New England Journal of Medicine, (1991) 324:1177-1184. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5104503/
[5] Antimatter particles have the same mass and spin as their matter counterparts but opposite electrical charges. For example, the electron (with a negative charge) has an antimatter partner, the positron, with a positive charge. When an electron and positron collide, they annihilate, converting entirely into energy, leaving a flash of light in their wake.
[6] Not all D sugars are sweet to taste. For example, D-mannose, which is D-sugar, is a non-sweet sugar. While it has the same chirality as glucose, its structure doesn’t interact well with sweetness receptors. Sweetness is perceived when a sugar molecule binds to specific taste receptors on our tongue, particularly the TAS1R2/TAS1R3 receptor complex. The shape and chemical properties of the sugar molecule determine how well it fits these receptors and triggers the sensation of sweetness. Beyond chirality (D or L), the specific structure and functional groups within a sugar molecule play a critical role in sweetness. For example, the presence of hydroxyl groups (-OH) and the arrangement of carbon atoms can influence sweetness. However, statistically, most D sugars and all common sugars are sweet.
[7] While bitterness is a common taste associated with some amino acids, others might have a different taste profile. For example, glutamic acid is the savory component of umami taste. However, most L-amino acids are tasteless, whereas D-forms tend to taste sweet. (Uwe J. Meierhenrich, Amino acids and the Asymmetry of Life, Berlin, Springer, 2008.) Interestingly, some L-amino acids can be both sweet and bitter, depending on the concentration. This complex taste perception highlights the intricate interaction between our taste receptors and the structure of molecules. It is important to note that the perception of bitterness can vary between individuals, and some people might not find all amino acids particularly bitter. Additionally, the context (other food components present) can also influence taste perception.
[8] It appears to me that a similar spiritual dynamic took place between Jacob and his wife. According to the Midrash, anticipating Laban’s trickery, Jacob and Rachel exchange a secret code before their wedding (Genesis Rabbah 70:19). On the wedding night, Rachel was to touch Jacob’s right earlobe, his right thumb, and the right big toe. However, at the last moment, not wanting to embarrass her sister, and unbeknownst to Jacob, Rachel revealed the code to Leah. As a result, was unaware of the substitute and marries Leah, only to later marry Rachel as well. According to Kabbalah, Jacob, who personified the partzuf Zeir Anpin (Z”A), was destined to have two wives. The reason is the female counterpart (and a “mate”) for the Z”A is partzuf Nukvah, which has two sub-partzufim—partzuf Leah and partzuf Rachel. Terrestrial Leah and Rachel were sisters, and, according to Midrash, they were twins (Genesis Rabbah 70:16). In other words, there was a symmetry between them, which made them interchangeable in a sense, allowing Laban to substitute Leah for Rachel (I wrote about this in my essay, “Entangled Sisters“). The code might have meant to break this symmetry to allow for differentiation between partzuf Leah and partzuf Rachel. So it seems to me, but I am not entirely sure that this is the correct interpretation.
